Great Epics Series
Among Heroes, Lu Bu Tales from the Romance of the Three Kingdoms
Among Heroes, Lu Bu; Among Horses, Red Hare
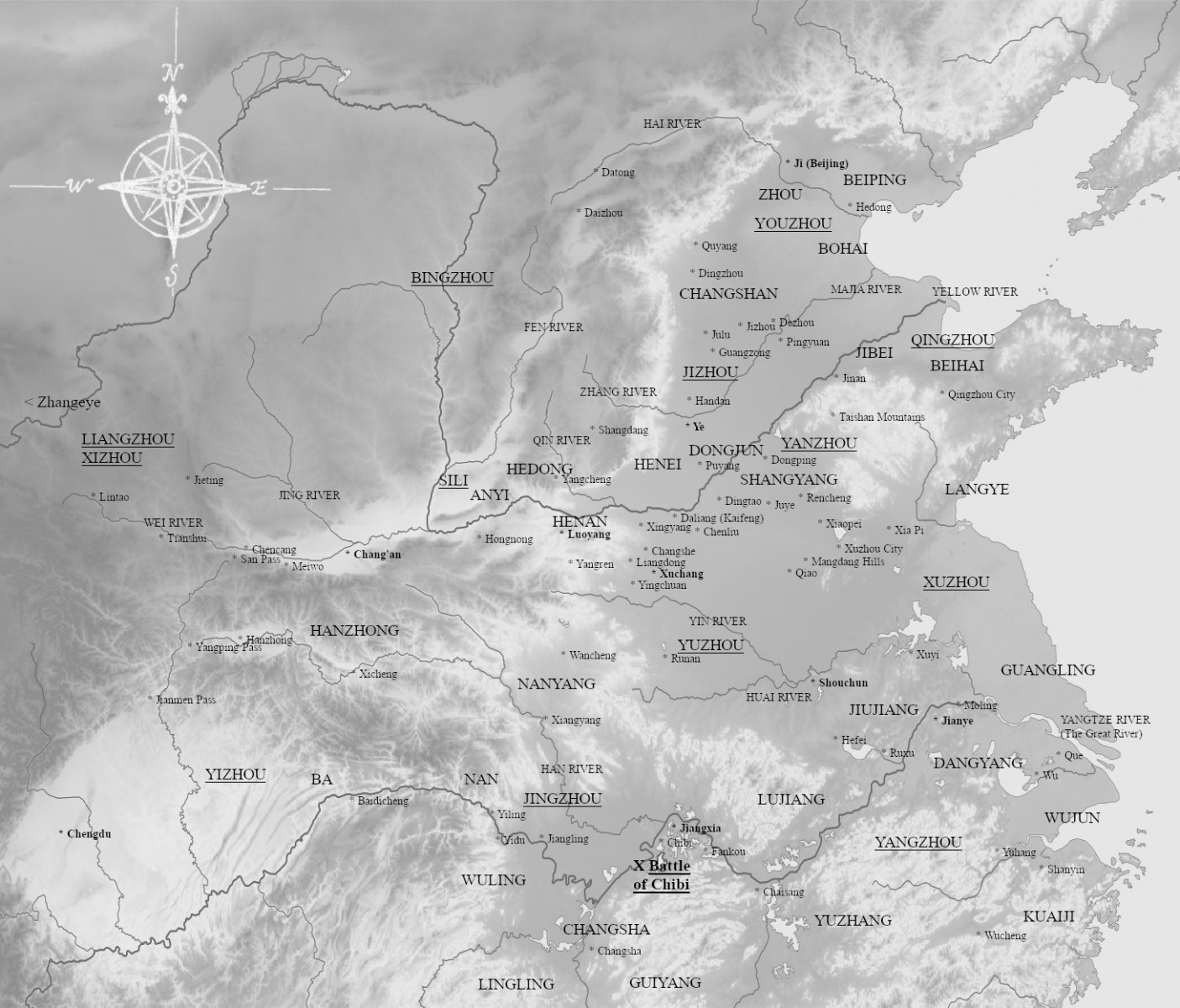
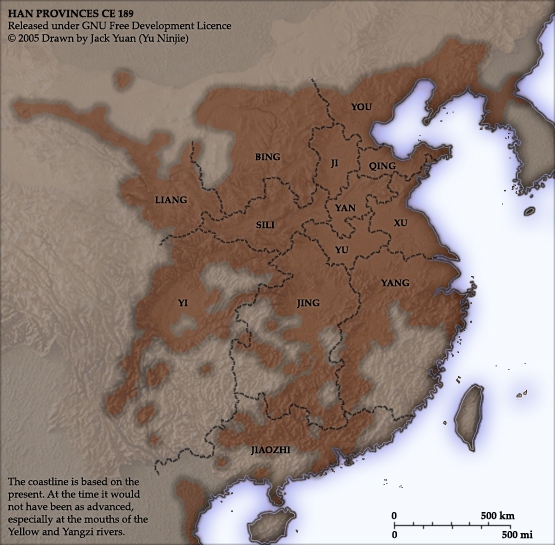
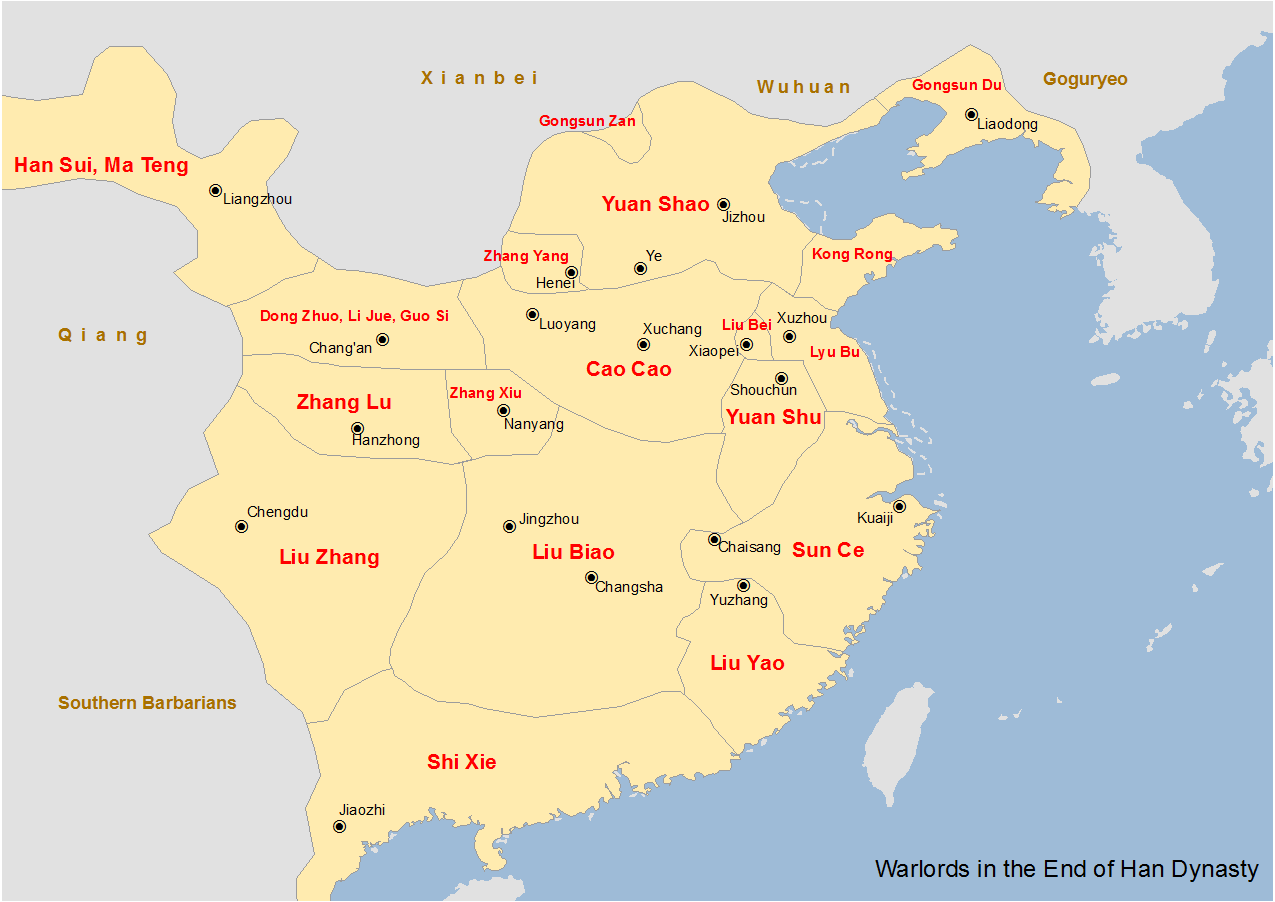
The Han Empire is falling. Mighty warlords raise up, vying for control to claim the ultimate glory of establishing China’s next great dynasty. It is a time of cunning villains and great heroes, of beautiful temptresses and brutal tyrants. One man stands above them all—Lu Bu, the mighty warrior, riding on Red Hare, the horse of legend that can run a thousand li. All who challenge him on the battlefield fall before his great halberd; his mighty bow has ended wars with a single shot. But will his might and courage alone be enough to win an empire and determine the future of all China?
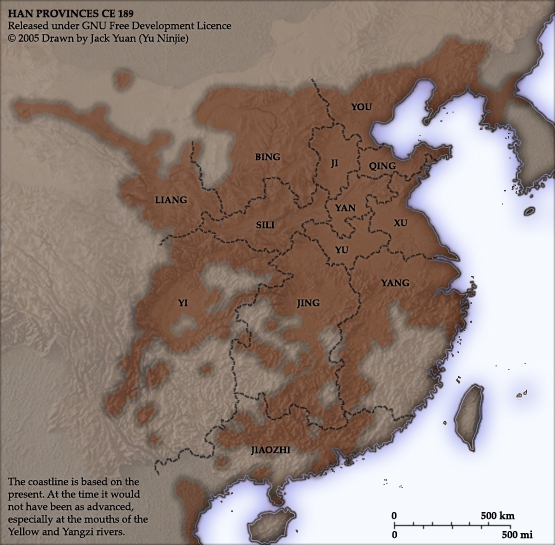
Romance of the Three Kingdoms is contributed to Luo Guanzhong in the Ming dynasty, and it has been a literary treasure to China for over 600 years. This historical epic is brought to vibrant life, so that you may enjoy the stories through the perspective of the original 14th century audience. This series is extracted from the full text edition translated by C. H. Brewitt-Taylor with modern Chinese spellings, augmented with over 200 historical illustrations, deep-dive commentaries, maps, and timelines to help the reader immerse themselves into the chaos of 2nd century China in the Age of Heroes of the Later Han and the Three Kingdoms period.